According to the European Regulation 1223/2009 and the National Legislation, cosmetic products shall be made available on the market only if the container and packaging bears the following information in indelible, easily legible and visible lettering:
(a) the name or registered name and the address of the responsible person. Such information may be abbreviated in so far as the abbreviation makes it possible to identify that person and his address. If several addresses are indicated, the one where the responsible person makes readily available the product information file shall be highlighted. The country of origin shall be specified for imported cosmetic products;
(b) the nominal content at the time of packaging, given by weight or by volume, except in the case of packaging containing less than five grams or five millilitres, free samples and single-application packs;
(c) the date until which the cosmetic product, stored under appropriate conditions, will continue to fulfil its initial function and remain in conformity with Article 3 (‘date of minimum durability’), or indication of the period after opening (PAO)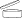
(d) particular precautions to be observed in use, and at least those listed in Annexes III to VI and any special precautionary information on cosmetic products for professional use;
(e) the batch number or the reference for identifying the cosmetic product. Where this is impossible for practical reasons because the cosmetic products are too small, such information need appear only on the packaging;
(f) the function of the cosmetic product, unless it is clear from its presentation;
(g) a list of ingredients. This information may be indicated on the packaging alone. The list shall be preceded by the term ‘ingredients’. All ingredients present in the form of nanomaterials shall be clearly indicated in the list of ingredients. The names of such ingredients shall be followed by the word ‘nano’ in brackets.
The information specified in points (b), (c) and (f) can be written in English or Greek without the prohibition of use of any additional language.
The information specified in point (d) must be written in Greek, without the prohibition of use of any additional language.